
**Using relative charges and masses are used to keep calculations simple. They have the same atomic number (same number of protons and electrons), but a different mass number due to more or fewer neutrons. Isotope is one of two or more forms of atoms of an element that differ in their number of neutrons. 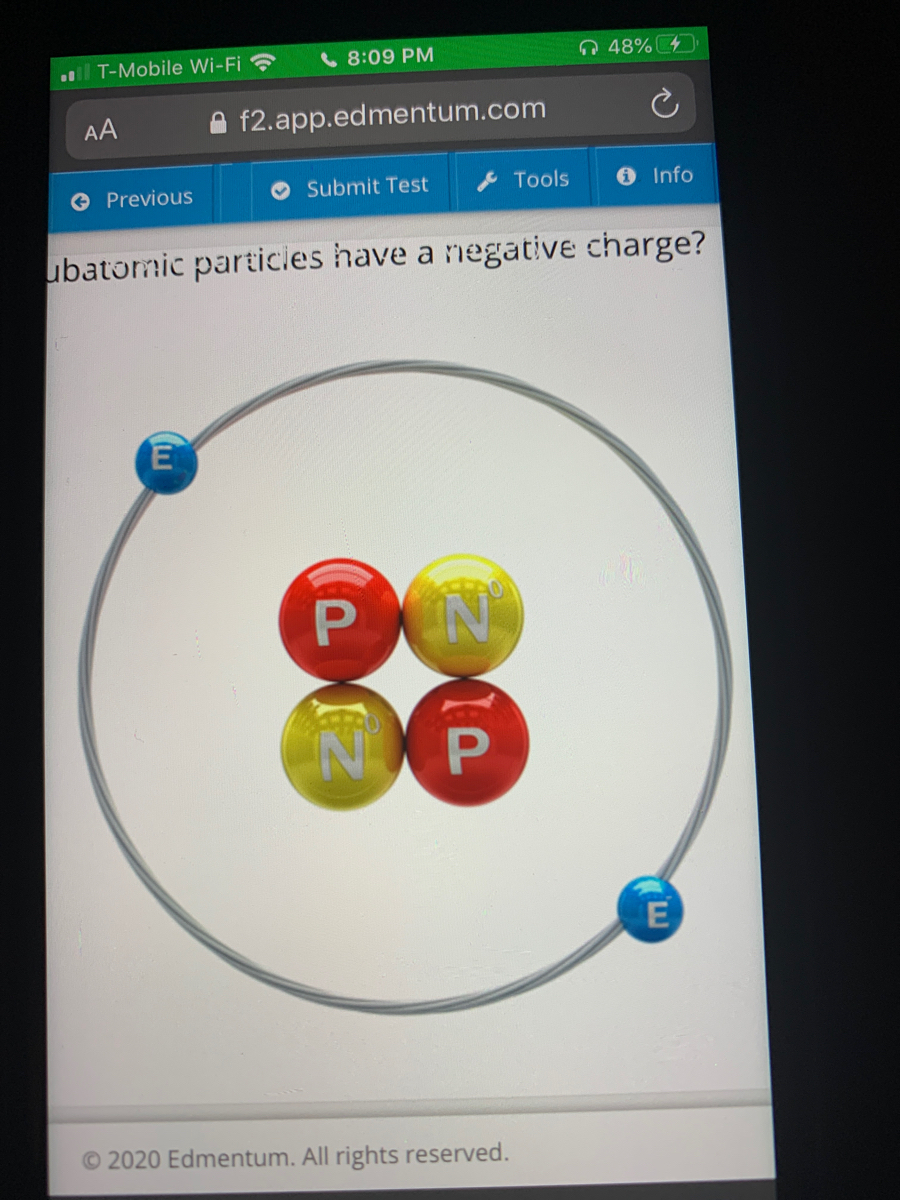
Discover the definitions of these subatomic particles with examples such as their mass. MCAT and Organic Chemistry Study Guides, Videos, Cheat Sheets, tutoring and. You will also learn about their location within the atom as well as mass and charge of each. The spacetime matrix, the Higgs field, is composed from. A subatomic particle is the umbrella term for protons, neutrons, and electrons. Subatomic Particles and Atomic Structure - This tutorial video will provide you with a brief review of the 3 main subatomic particles including protons, neutrons and electrons. For a given element, the mass number is the number of protons and neutrons (nucleons) in the nucleus. The subatomic particles are manifestations of their quantum fields formed as geometrical excitations within the local spacetime matrix. Neutron is an uncharged particle of the nucleus of all atoms EXCEPT hydrogen. Ion is an atom that has gained or lost one or more electrons, thus becoming positively or negatively charged. Electrons can be shared or transferred among atoms.Ītoms have an equal number of protons and electrons therefore, they have a no net charge. any Chlorine atom is going to have 17 electrons). Each subatomic particle has a relative mass. 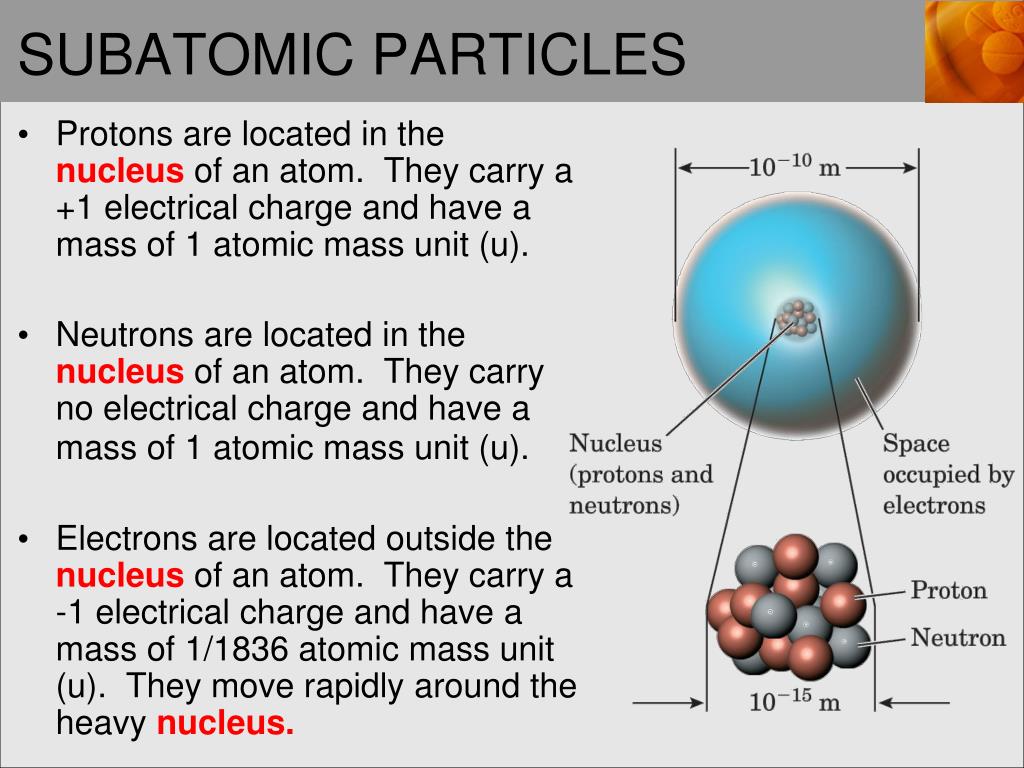
Protons and neutrons are located in the nucleus, a dense central core in the middle of the atom, while the. The last column in the table lists the location of the three subatomic particles. All atoms of an element have the same number of electrons (i.e. Atoms are made of protons, neutrons, and electrons, which are types of particle also known as subatomic particles. The answer is that there are equal numbers of protons and electrons equal numbers of positive and negative charges so they cancel each other out. The atomic number of an element represents the number of protons in the nucleus.Įlectron (e -) is negatively charged particle that can occupy a volume of space (orbital) around an atomic nucleus. Proton (p +) is positively charged particle of the atomic nucleus.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
